PRISM: A Reproducible Framework for Systematic Pharma Pricing — Product Equivalence, Right-to-
Price, Pack Architecture, and Elasticity-Based Optimization
Every global pharma needs a pharma pricing engine — a single, reproducible system for price realization across countries, brands, channels, and pack configurations. Pharma pricing leaders already own the analyses. A country team can build a workbook. A regional pricing lead can pull a competitive benchmark. A global team can ask for a Right-to-Price view. The harder question is whether the same company can answer the same question again next cycle — across countries, brands, channels, pack configurations, currencies, competitors, and regulatory constraints — with an audit trail a pricing committee can defend. PRISM is the pharma pricing engine for installing that capability.
Why Reproducibility Matters More Than Analytics
Price realization rarely leaks in one obvious place. It leaks through the seams of the operating model: net price reconstructed manually from ERP, finance accruals, and channel files; competitor products compared at pack level when strength or pack size differs; product equivalence maintained as workbook tabs rather than a logic layer; pack architecture drifting silently across years of inflation moves; elasticity applied after a price action has already shipped.
The result is not a lack of pricing strategy — it is an operating-model failure. The company owns many analyses but does not own a reproducible pharma pricing engine. In Deloitte’s 2025 Life Sciences Outlook, 47% of C-suite executives expected pricing and access to significantly affect their 2025 strategies, with another 49% expecting moderate impact. The pressure on the capability is structural.
What You Will Learn
The 53-page whitepaper walks through the framework module-by-module with worked examples, anonymized platform illustrations, and the architectural decisions that make the engine reproducible:
- DDD-Equivalized Competitive Pricing (Module 1): How to normalize molecule, strength, form, route, and pack into a Defined Daily Dose comparison — and read the competitive gap at the only layer where the comparison is honest.
- Right-to-Price and Value-Based Positioning (Module 2): A seven-dimension scoring rubric (brand equity, differentiation, perceived worth, market stability, supply reliability, access position, competitor alternatives) that converts brand worth into a defensible price band.
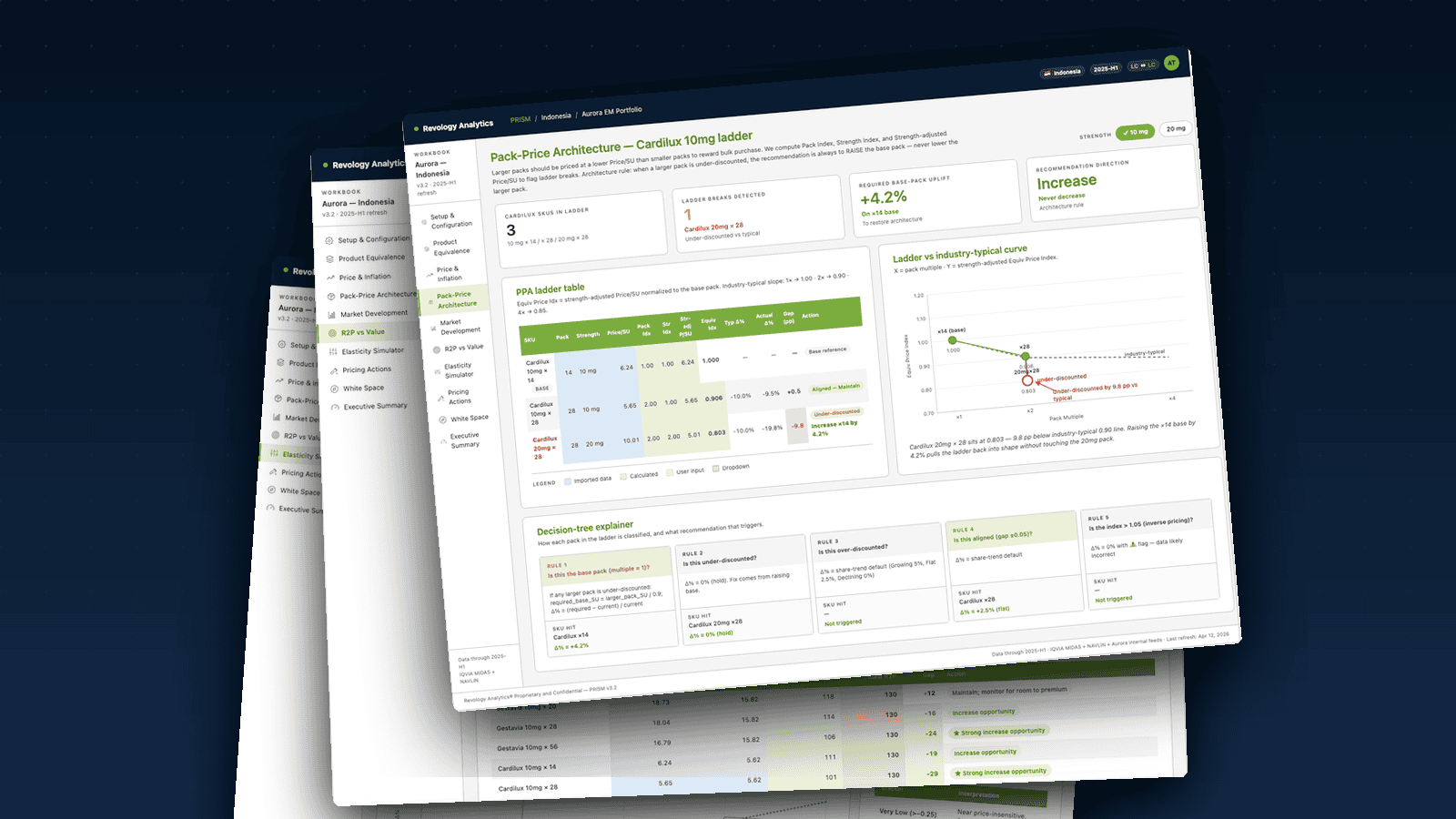
- Price Pack Architecture (Module 3): Why pack ladders decay, how the pack-index / strength-index / equivalent-price-index logic surfaces it, and the architectural rule — raise the base, never lower the larger pack — that protects revenue.
- Elasticity Simulator (Module 4): How to size every recommendation against modeled own-price elasticity with a cross-elasticity guardrail — and why a credible engine must be willing to return no action when the economics don’t support a move.
Designed for Pharma Pricing, Commercial, and Executive Leaders
The whitepaper offers practical frameworks for three pharma-leadership archetypes:
- Pricing, RGM, and Market Access leaders: Methodology depth your team will adopt — the Product Equivalence Matrix, DDD assignment logic, R2P rubric, pack-architecture rule set, elasticity discipline, and the five-phase deployment sequence that installs the engine without asking country teams to abandon Excel.
- Commercial and Brand leadership: One ranked action list, not ten disconnected analyses — how the engine sequences DDD competitive gaps, R2P drift, pack-ladder distortions, and elasticity into SKU-level recommendations labeled increase, maintain, monitor, or hold.
- C-suite (CCO, CFO, CEO): The operating-model shift from workbook-led to engine-led pricing, the five-level pharma pricing maturity model, and the ten-question executive checklist for diagnosing workflow fragility before investing in another tool.
Key Insights You’ll Gain
- The four-module composition logic. Why DDD-equivalized pricing, Right-to-Price, pack architecture, and elasticity become more valuable when they run together than when they run alone.
- Anonymized evidence from four pharma market archetypes. Mid-single to low-double-digit % value pools, with 5–20% net price realization across the pilot — including the largest single finding of approximately $6.3M on a pack-architecture distortion (figures 2× scaled per the anonymization protocol).
- The six principles of a reproducible pricing engine. Single data contract, templates as code, deterministic transforms, scenarios separated, versioned outputs, and a human-in-the-loop review gate.
- The five-level pharma pricing maturity model. From ad-hoc workbook to continuous pricing intelligence — and how to identify the highest-value seam to start with.
- The ten-question executive checklist. A practical diagnostic any pricing leader can run against their own workflow in an afternoon.
Take Action: Install the Pharma Pricing Engine
In a regulated, multi-country pharma environment, mastering pricing reproducibility gives your team the capability to defend every committee recommendation — across cycles, currencies, regulators, and committees. Download the complete whitepaper to see the full framework, the platform illustrations, and the evidence from the anonymized pilot.
Download Now to receive the 53-page whitepaper, the companion one-page Pharma Pricing Reproducibility Checklist, and a short three-part follow-up over ten days from Armin Kakas covering the module pharma teams most often skip — and what it costs them.